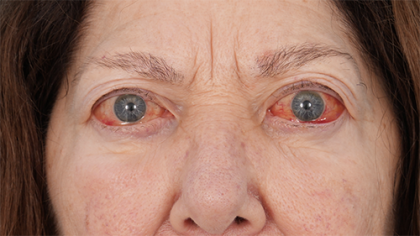
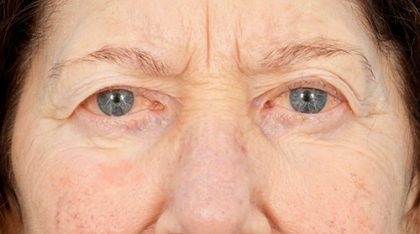
Tepezza Before And After: Real Patient Transformations & What To Truly Expect
Have you ever scrolled through medical forums or patient support groups and wondered, "What do real Tepezza before and after results actually look like?" The curiosity is completely understandable. For those living with the frustrating and often painful symptoms of Thyroid Eye Disease (TED), the promise of a treatment that can reverse eye bulging, double vision, and inflammation feels almost too good to be true. Tepezza (teprotumumab-trbw) didn't just enter the market; it revolutionized the treatment landscape for TED. But beyond the clinical trial headlines and FDA approval announcements, what does the journey from "before" to "after" genuinely entail? This article dives deep into the tangible, day-to-day realities of Tepezza treatment. We’ll move beyond the marketing to explore the nuanced timeline of changes, the challenges during the process, and the inspiring real-world transformations that offer genuine hope to patients.
Thyroid Eye Disease is an autoimmune condition where the tissues and muscles behind the eyes become inflamed and swollen. This "before" state is more than just a cosmetic concern; it's a life-altering reality. Patients often describe feeling like a stranger in their own reflection, struggling with eyes that feel pushed forward (proptosis), constantly irritated, or misaligned causing persistent diplopia (double vision). Simple tasks like applying makeup, reading, or driving become arduous. The psychological toll is significant, with many reporting anxiety, social withdrawal, and depression. Understanding this starting point is crucial because it frames the magnitude of change Tepezza aims to achieve. The goal isn't merely aesthetic improvement; it's about restoring function, comfort, and quality of life.
Understanding Thyroid Eye Disease (TED) and the "Before" State
Before we can appreciate the "after," we must fully grasp the challenging "before." Thyroid Eye Disease, also known as Graves' Orbitopathy or Graves' Ophthalmopathy, is a complex condition intricately linked to thyroid dysfunction, though it can occur with normal thyroid function. The immune system mistakenly attacks the tissues around the eyes, causing inflammation, swelling, and the buildup of glycosaminoglycans (GAGs). This buildup pushes the eye forward into the socket and can tighten the muscles that control eye movement.
The symptoms of active TED can vary in severity but typically include:
- Proptosis (Bulging Eyes): The most visible sign, measured in millimeters by an ophthalmologist.
- Periorbital Edema (Swelling): Puffy eyelids and tissue around the eyes.
- Diplopia (Double Vision): Caused by inflamed and scarred eye muscles that restrict movement.
- Ocular Irritation: A gritty, burning sensation, excessive tearing, or light sensitivity due to the eyes being more exposed.
- Vision Changes: In severe cases, compression of the optic nerve can lead to vision loss—a medical emergency.
This "before" phase is the active, inflammatory stage. It's a period of uncertainty where symptoms can worsen over weeks or months. Patients often feel powerless, watching their appearance and vision change with limited treatment options beyond high-dose steroids (which have significant side effects) or drastic surgical interventions reserved for the chronic, "burnt-out" phase. The emergence of Tepezza offered a new hope specifically for this active phase, targeting the disease at a molecular level to halt progression and reverse damage.
What Is Tepezza and How Does It Work?
Tepezza is the first and only FDA-approved medication specifically for the treatment of Thyroid Eye Disease. Its generic name is teprotumumab-trbw, and it’s a monoclonal antibody. This means it’s a lab-created protein designed to target a specific molecule in the body. Tepezza works by inhibiting the Insulin-like Growth Factor-1 Receptor (IGF-1R).
- Pineapplebrat Nudes
- Reagan Gomez Prestons Shocking Leak The Video That Destroyed Her Career
- The Secret Sex Tape Everyones Talking About Michelle Myletts Leaked Scandal Exposed
Here’s why that’s so important: In TED, the IGF-1R and the Thyroid Stimulating Hormone Receptor (TSHR) form a functional unit on the surface of the cells behind the eyes (fibroblasts and preadipocytes). When thyroid antibodies activate the TSHR, it also activates the IGF-1R, triggering a cascade that leads to the production of those problematic GAGs and inflammation. By blocking the IGF-1R, Tepezza disrupts this entire signaling pathway. It effectively tells the cells, "Stop producing the inflammatory substances and stop swelling." It doesn't just mask symptoms; it addresses the underlying autoimmune activity driving the disease.
The treatment protocol is an intravenous (IV) infusion. The standard regimen is an initial dose, followed by infusions every 3 weeks for a total of 8 doses over about 6 months. This structured timeline is critical to understanding the Tepezza before and after journey. The effects are not instantaneous; they build as the medication accumulates in the system and gradually reduces inflammation and swelling.
The Tepezza Timeline: What to Expect Week by Week
One of the most common questions is, "How quickly will I see Tepezza before and after changes?" The answer is nuanced and varies by individual, but clinical trials and patient reports provide a clear general pattern. It’s a marathon, not a sprint, with improvements unfolding in stages.
Weeks 1-4 (Infusions 1-2): The "Quiet" Phase. During the first month, patients are often focused on managing initial side effects like hyperglycemia (elevated blood sugar), nausea, fatigue, or muscle cramps. Visually, changes are usually minimal or not yet noticeable to the patient. However, the medication is actively working at the cellular level to dampen the inflammatory process. Patience is paramount here. It’s easy to feel discouraged, but this foundational work is essential for the transformations to come.
Weeks 5-12 (Infusions 3-5): The First Glimpses of Change. This is when many patients start to see the first tangible "after" signs. Reduction in periorbital swelling (eyelid edema) is often the earliest visible change. The eyes may start to look less puffy. Some patients also report a decrease in the gritty, foreign-body sensation. Proptosis (bulging) may begin a very gradual reduction, but it’s often subtle at this stage. The improvement in diplopia (double vision) can be more noticeable for some, as muscle inflammation decreases and eye alignment begins to improve. This phase builds crucial motivation.
Weeks 13-24 (Infusions 6-8): The Transformation Phase. This is the period of the most dramatic Tepezza before and after results. Clinical trial data showed that by 24 weeks, the average reduction in proptosis was 3.2 millimeters. For a patient, this can mean the difference between eyes that feel and look "stuck out" and a much more natural, recessed position. Double vision often continues to improve or may resolve completely. The overall inflammation subsides, leading to less redness and irritation. Patients frequently describe looking in the mirror and seeing a version of themselves they haven't seen in years. The final infusion at week 24 marks the end of the active treatment phase, but the improvements often continue for several months afterward as the body stabilizes.
Navigating Side Effects: The "During" Phase
A complete picture of the Tepezza before and after experience must include the "during"—the management of side effects. While transformative, Tepezza is a potent medication, and its adverse events are a significant part of the journey. Being prepared is key to adherence and success.
The most common side effects, based on clinical trials, include:
- Hyperglycemia: Elevated blood sugar levels, particularly concerning for patients with pre-existing diabetes or prediabetes. This requires proactive monitoring. Patients are advised to check blood glucose frequently, often before each infusion, and work closely with their endocrinologist or primary doctor to manage diet, exercise, and potentially adjust diabetes medications.
- Hearing Issues: Tinnitus (ringing in the ears) and hypoacusis (hearing loss) have been reported. Audiograms (hearing tests) are typically recommended before starting treatment and periodically during. Any new or worsening symptoms should be reported immediately.
- Infusion Reactions: Symptoms like shortness of breath, rash, or chest pain during the infusion. Infusions are administered in a medical setting where these can be managed.
- Gastrointestinal Issues: Nausea, diarrhea, and abdominal pain.
- Fatigue and Muscle Cramps: Often manageable with rest and hydration.
Actionable Tips for Managing Side Effects:
- Build Your Care Team: Have an endocrinologist/primary doctor and an ophthalmologist (specifically a neuro-ophthalmologist or orbital specialist) in close communication.
- Monitor Diligently: Keep a log of blood sugar readings, any hearing changes, and other symptoms.
- Hydrate and Nourish: Drink plenty of water and eat balanced meals to help manage nausea and support overall health.
- Communicate Openly: Never hesitate to tell your infusion nurse or doctor about any discomfort. They can often adjust pre-medications (like anti-nausea drugs or steroids) to improve tolerance.
- Plan for Rest: Schedule infusion days with minimal other obligations. Fatigue is common in the 24-48 hours post-infusion.
Real Tepezza Before and After: Patient Stories & Clinical Data
The numbers from the Phase 3 clinical trials are compelling: 83% of Tepezza-treated patients achieved a reduction in proptosis of 2mm or more compared to 10% on placebo. 69% experienced an improvement in diplopia (double vision) versus 29% on placebo. These statistics represent real people, but individual stories bring the data to life.
Consider "Sarah," a 45-year-old teacher with active TED for 18 months. Her "before" was defined by constant eye pressure, severe dryness requiring hourly artificial tears, and proptosis measuring 24mm in her right eye and 23mm in her left. Double vision made reading lesson plans exhausting. Her Tepezza before and after journey saw her first noticeable change at infusion #4: her eyelids were less puffy. By infusion #6, her proptosis measurements had dropped to 22mm and 21mm. Most importantly, her diplopia resolved completely after infusion #7. At her 6-month post-treatment visit, she reported being able to read without strain and feeling confident making eye contact with her students again.
Or "David," a 52-year-old marketing executive. His "before" included significant facial swelling that made him look perpetually tired and angry. He also had mild optic nerve compression threatening his peripheral vision. His transformation was slower but profound. The periorbital edema resolved first, then the proptosis gradually decreased. His most significant "after" was the stabilization and improvement in his visual field test, which his doctor confirmed was directly linked to the reduction in orbital inflammation and pressure from Tepezza.
These stories align with the clinical data: improvement is the norm, but the pace and specific sequence of changes are personal. Some see swelling resolve first; others get quicker relief from double vision. The key is consistent treatment and management of side effects.
Tepezza vs. Other TED Treatments: How Does It Compare?
To fully understand the Tepezza before and after paradigm shift, it helps to contrast it with previous treatment options, which were largely reactive or surgical.
- High-Dose Steroids (e.g., Methylprednisolone): The old first-line for active, moderate-to-severe TED. Steroids reduce inflammation quickly but do not address the underlying IGF-1R pathway. Their effects are often temporary, and the side effect profile (weight gain, mood swings, diabetes, osteoporosis) is notoriously harsh, especially for long courses. Tepezza offers a targeted biological approach with a different, though still significant, side effect profile focused on metabolic and auditory systems.
- Orbital Radiotherapy: Used for active inflammation, often combined with steroids. Its mechanism is different and its efficacy is debated, with potential long-term risks. It’s not a targeted therapy like Tepezza.
- Surgical Interventions (Orbital Decompression, Strabismus Surgery, Eyelid Surgery): These are definitive, corrective surgeries but are only recommended once the disease has been inactive and stable for at least 6-9 months. They are irreversible and address the consequences of TED (bone removal to make space, muscle adjustment) but not the active inflammation. Tepezza can potentially reduce the need for extensive decompression surgery by shrinking the swollen tissues before they cause permanent bony changes.
In essence, Tepezza fills a critical gap: a pharmacological treatment for the active inflammatory phase that can alter the disease's natural history. It’s not a cure—the underlying autoimmune propensity remains—but it can push a patient from active disease into a stable, inactive state with dramatically improved appearance and function, often avoiding or minimizing the need for later surgery.
Key Questions Answered: Tepezza Before and After Concerns
Q: Are Tepezza results permanent?
A: The results from a full course of Tepezza are generally long-lasting, with many patients maintaining improvements for years. However, TED is a chronic condition. There is a possibility of disease recurrence or new inflammatory activity in the future, though clinical trial follow-up data shows low recurrence rates. Long-term monitoring with an orbital specialist is essential.
Q: How much does Tepezza cost, and is it covered by insurance?
A: The list price is substantial (tens of thousands of dollars for the full course). However, most major commercial insurers and Medicare now provide coverage for FDA-approved indications (active, moderate-to-severe TED). The prior authorization process can be complex, requiring detailed documentation from your specialist (ophthalmology notes, imaging, clinical severity scores). Patient support programs from the manufacturer (Genentech) offer co-pay assistance for eligible commercially insured patients.
Q: What if my "after" isn't as dramatic as the clinical trials?
A: Not every patient achieves a 3mm+ reduction in proptosis. Response is variable. The goal is clinically meaningful improvement, which can be defined as reduced swelling, improved eye movement, pain relief, and prevention of worsening. Even a 1-2mm reduction can significantly improve appearance and comfort. It’s vital to have realistic expectations discussed thoroughly with your doctor before starting treatment.
Q: Can Tepezza be used for cosmetic reasons alone?
A: No. Tepezza is strictly approved for the treatment of the active, inflammatory phase of Thyroid Eye Disease with clinical signs like proptosis, diplopia, or inflammation. It is not a cosmetic treatment for mild, inactive, or age-related bulging eyes. Using it outside its approved indication is not only ineffective but also ethically and legally problematic.
Conclusion: The Realistic Promise of the Tepezza Before and After Journey
The narrative of Tepezza before and after is ultimately a story of medical progress meeting human experience. It’s a story that begins with the frustration of an active autoimmune assault on the eyes and, for many, culminates in a profound reduction of that assault—a return to a more familiar reflection and a more comfortable, functional vision. The journey requires commitment: to the 8-infusion schedule, to vigilant management of side effects, and to patience as the body heals.
The "after" is not a mythical, perfect restoration. It is a realistic, often dramatic, improvement. It is the ability to apply mascara without your brush hitting your brow, to read a book without a headache, to make direct eye contact without self-consciousness. It is the relief of inflammation subsiding and the halting of progressive damage. The Tepezza before and after transformation is a powerful testament to targeted biological therapy. If you or a loved one is navigating the challenging "before" of Thyroid Eye Disease, consulting with an experienced orbital specialist about whether Tepezza is a suitable option is the most critical first step. This treatment represents a monumental shift, offering a beacon of hope where once there were few active-phase options. The real-world results, supported by both rigorous science and countless patient journeys, confirm that for many, a meaningful and life-changing "after" is truly possible.