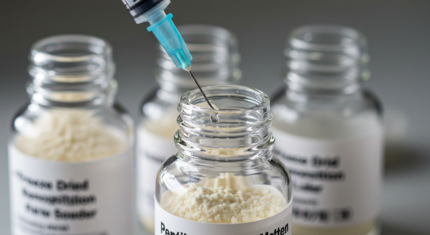
How To Reconstitute Peptides: A Complete Step-by-Step Guide For Beginners
Have you ever received a vial of lyophilized (freeze-dried) peptide powder and wondered, "How do I actually reconstitute this correctly?" You're not alone. For researchers, clinicians, and informed enthusiasts, mastering the art of peptide reconstitution is a critical first step. A single mistake can render a valuable, often expensive, compound ineffective or even dangerous. This comprehensive guide will walk you through every detail, transforming you from a hesitant beginner into a confident practitioner. We’ll cover everything from the non-negotiable rules of sterility to the precise math behind dilution ratios, ensuring your peptides are potent, pure, and safe for their intended application.
Why Proper Reconstitution is Non-Negotiable: More Than Just Adding Water
Peptides are delicate chains of amino acids. In their lyophilized state, they are stable and have a long shelf life. The moment you introduce a solvent, you initiate a biochemical process. Incorrect reconstitution can lead to rapid degradation, bacterial contamination, and inaccurate dosing. Think of it like reviving a dormant seed; you need the right environment (solvent), the right amount of water (volume), and sterile conditions to prevent mold. The consequences of poor technique range from wasted research funds to compromised experimental results and potential health risks if used therapeutically. According to best practices in laboratory medicine, maintaining aseptic technique is paramount, as even a single bacterial cell can multiply exponentially in a nutrient-rich peptide solution. This foundational understanding sets the stage for the meticulous process that follows.
The Golden Rule: Mastering Sterile Technique and Your Workspace
Before you even touch the vial, your environment must be pristine. This is the single most important factor in preventing contamination.
- Cookie The Monsters Secret Leak Nude Photos That Broke The Internet
- Leaked Mojave Rattlesnakes Secret Lair Found You Wont Believe Whats Inside
- Ward Bonds Secret Sex Tape Leaked Hollywoods Darkest Hour Exposed
Creating Your Sterile Field
Your workspace, whether a dedicated lab bench or a clean home office area, must be disinfected. Use 70% isopropyl alcohol to wipe down all surfaces—your bench, pipettes, vial stoppers, and any tools. Allow it to air dry completely; wiping it can reintroduce contaminants. Work in a still-air environment. Drafts from fans, open windows, or movement can carry airborne microbes. Many serious practitioners use a laminar flow hood or a still-air box, which provides a constant stream of filtered air. If that's not available, choose the cleanest, quietest room possible and minimize movement.
Personal Hygiene and Gloving
Wash your hands thoroughly with soap and water for at least 20 seconds, then dry them with a lint-free towel. Some experts recommend using an alcohol-based hand rub after washing. Always wear powder-free nitrile gloves. Powder can contaminate your solution, and latex can cause allergic reactions that may interfere with some assays. Change gloves if they become soiled or torn, and consider changing them between handling different vials to prevent cross-contamination. Avoid touching your face, hair, or any non-sterile surface while gloved.
Tool Sterilization
All tools that will contact the peptide or solvent must be sterile. This includes:
- Syringes and Needles: Use sterile, single-use, disposable syringes and needles. Never reuse.
- Pipettes and Tips: If using a micropipette, ensure the tip is sterile and aerosol-resistant. Use a new tip for every transfer.
- Vial Stoppers: The rubber stopper on your peptide vial is a potential contamination point. Swab it thoroughly with an alcohol pad, let it dry, and only pierce it with your sterile needle/syringe. Never touch the needle tip to any non-sterile surface.
Choosing the Right Solvent: Not All Water is Created Equal
The choice of solvent is your first major decision and depends entirely on the peptide's sequence and intended use.
Bacteriostatic Water (0.9% Benzyl Alcohol)
This is the most common and generally recommended solvent for reconstitution. The small percentage of benzyl alcohol acts as a preservative, inhibiting bacterial growth once the vial is opened and stored in the refrigerator. It is ideal for peptides that will be used over several days or weeks. Important: Benzyl alcohol can be irritating to some tissues and is generally not suitable for injectable use in certain applications or for individuals with sensitivities. Always check the manufacturer's guidelines.
Sterile Water for Injection (SWFI)
This is pure, pyrogen-free water with no additives. It is the preferred solvent for injectable peptides intended for immediate use or where any additive is contraindicated. Because it lacks a preservative, any solution reconstituted with SWFI must be used within a very short timeframe (often 24-48 hours) and stored meticulously in the refrigerator. It is also suitable for peptides destined for in vitro (cell culture) applications where benzyl alcohol might be toxic to cells.
Acetic Acid or Other Diluents
Some hydrophobic (water-fearing) peptides, particularly those with complex sequences, may have poor solubility in water alone. Manufacturers will often specify a dilute solution of acetic acid (e.g., 5-10%) or sometimes a small amount of ammonium hydroxide to help dissolve the peptide fully. Always follow the manufacturer's specific instructions first. If none are given, a general rule is to start with bacteriostatic water and, if dissolution is slow or incomplete, add a drop of glacial acetic acid to the solvent vial, mix, and try again.
The Mathematics of Precision: Calculating Your Reconstitution Ratio
This is where many newcomers get anxious, but the math is straightforward. You are simply deciding on your target concentration (e.g., 1 mg/mL) and calculating the volume of solvent needed.
The Core Formula
Volume of Solvent (mL) = Peptide Mass (mg) / Desired Concentration (mg/mL)
Example: You have a 5 mg vial of peptide and you want a final concentration of 2 mg/mL.Volume = 5 mg / 2 mg/mL = 2.5 mL
You would add 2.5 mL of your chosen sterile solvent to the vial to achieve a 2 mg/mL solution.
Practical Application and Common Targets
- 1 mg/mL: Very common, easy to calculate. For a 2 mg vial, add 2 mL solvent.
- 2 mg/mL: A good middle ground, slightly more concentrated.
- 10 mg/mL: Used for very potent peptides where you need to draw a very small volume for a dose. Requires extreme precision in measurement.
Pro Tip: Use a reconstitution calculator (many are available online) to avoid arithmetic errors. Write down your calculation before you begin. Remember, the total volume in the vial after reconstitution is the solvent you added. The peptide powder itself has a small volume, but it is negligible and ignored in this calculation.
Step-by-Step Reconstitution Procedure
- Prepare: Have your solvent vial, peptide vial, alcohol pads, and sterile syringe/needle ready on your clean, disinfected workspace.
- Swab: Clean the tops of both the solvent vial and peptide vial with separate alcohol pads. Let them air dry.
- Draw Solvent: Draw the exact calculated volume of solvent into your sterile syringe. Expel any air bubbles by tapping the syringe and gently pushing the plunger until a tiny droplet appears at the needle tip.
- Inject Slowly: Insert the needle through the center of the peptide vial's rubber stopper at a 45-90 degree angle. Slowly inject the solvent down the side of the vial or onto the glass wall. Do not blast it directly onto the peptide cake. This prevents foaming and potential peptide damage.
- Dissolve: Gently swirl the vial. Do not shake. Shaking can denature (unfold and damage) the peptide structure. Swirl until the powder is completely dissolved. This may take a few minutes. If stubborn, let it sit for 5-10 minutes and swirl again. If using an acid solvent, dissolution is usually very rapid.
- Inspect: The solution should be clear and colorless (or slightly tinted if an acid was used). Any cloudiness, particles, or discoloration indicates contamination or incomplete dissolution—do not use it.
- Label Immediately: Write the peptide name, concentration (e.g., 2 mg/mL), date of reconstitution, and solvent used (e.g., "Bac Water") on the vial with a permanent, solvent-resistant marker.
Storage and Stability: Protecting Your Investment
Reconstituted peptides are far less stable than their lyophilized form. Proper storage is critical for maintaining potency.
Temperature is Key
- Refrigeration (2-8°C / 35-46°F): This is the standard for most reconstituted peptides stored in bacteriostatic water. It significantly slows bacterial growth and chemical degradation. Always store in the refrigerator, not the freezer. Freezing can cause precipitation and denaturation.
- Freezing (-20°C): Generally not recommended for reconstituted peptides due to ice crystal damage. However, some very stable peptides may be frozen in single-use aliquots. Only do this if explicitly stated by the manufacturer.
- Room Temperature: Avoid. Degradation accelerates rapidly. A reconstituted peptide left at room temperature for more than a few hours should be considered compromised.
Light and Aliquoting
Many peptides are light-sensitive. Store vials in the dark, either in a closed drawer or wrapped in aluminum foil. To prevent repeated temperature fluctuations from opening a main vial, aliquot your reconstituted peptide into smaller, single-use volumes. Use sterile, low-binding microcentrifuge tubes. Label each aliquot with the same details. This way, you only remove one aliquot from the fridge for a single use, keeping the master stock stable.
Shelf Life: A General Guideline
- In Bacteriostatic Water, Refrigerated: Typically 7-30 days. Some highly stable peptides may last longer. When in doubt, use within 7-10 days.
- In Sterile Water for Injection, Refrigerated:24-48 hours maximum.
- Always consult the manufacturer's Certificate of Analysis (CoA) or technical data sheet for the specific peptide's stability data. This is your most reliable source.
Safety First: Handling and Disposal Protocols
Peptides are bioactive compounds. Treat them with respect.
Personal Protective Equipment (PPE)
At a minimum, wear gloves and safety glasses. If you are handling large quantities or peptides with unknown toxicity profiles, consider a lab coat. If there's a risk of aerosol generation (e.g., during vigorous mixing), work in a fume hood.
Avoiding Contamination and Exposure
Never recap a used needle. Dispose of it immediately in a sharps container. If a spill occurs, disinfect the area with an appropriate solution (e.g., 10% bleach followed by 70% alcohol). For skin contact, wash immediately with soap and water. For eye contact, flush with water for 15 minutes and seek medical attention.
Disposal
Reconstituted peptide solutions are considered biohazardous waste due to potential bacterial growth. Do not pour them down the sink. Dispose of them according to your local regulations for liquid chemical/biohazard waste. Typically, this involves placing the solution in a designated biohazard bag with solidifying agent or adding a disinfectant (like bleach) before disposal. Your institution's safety officer is the best resource for specific protocols.
Troubleshooting: What To Do When Things Go Wrong
Even with careful technique, issues can arise.
"The peptide won't dissolve!"
- Cause: The peptide may be highly hydrophobic, or the solvent is incorrect.
- Solution: First, ensure you used the correct solvent as per instructions. Gently warm the vial in your hand or a warm water bath (not hot!) for 5-10 minutes. Swirl frequently. If it remains cloudy, you may need to add a drop of dilute acetic acid (5-10%) to the vial, swirl, and reassess. Do not use sonication (ultrasonic bath) unless the manufacturer explicitly approves it, as it can severely damage peptides.
"The solution is cloudy or has particles after dissolving."
- Cause: This is almost always contamination (bacterial or fungal) or precipitation due to incorrect pH/solvent.
- Solution:Discard the vial immediately. Do not attempt to filter it, as this does not guarantee sterility and may adsorb peptide onto the filter. Review your sterile technique steps. Start over with a new vial, ensuring every step is executed flawlessly.
"I think I overdiluted/underdiluted."
- Cause: Error in the initial calculation or solvent measurement.
- Solution: If you realize the mistake immediately, you may be able to correct it. For example, if you added too much solvent (under-concentrated), you cannot easily remove solvent without risking contamination. The vial is likely ruined. If you added too little (over-concentrated), you can carefully add the correct additional amount of solvent, but this introduces a contamination risk. Prevention is key: double-check your math and syringe measurements before injecting the solvent.
Applications and Best Practices: From Lab to Clinic
Understanding how to reconstitute is only half the equation; knowing why and for what informs your technique.
Research Applications
In cell culture experiments, peptide concentration and sterility are critical. Contaminants can kill cell lines and invalidate months of work. For in vitro binding assays, inaccurate concentration leads to meaningless IC50 or EC50 values. Here, using SWFI and using the solution immediately is often the protocol.
Therapeutic and Clinical Use
For subcutaneous or intramuscular injections (common for peptides like BPC-157, Thymosin Alpha-1), sterility and pyrogen-free status are absolute requirements. Bacteriostatic water is standard due to its multi-use nature, but the user must be scrupulous with vial handling. Never share needles or vials. Dosing accuracy is paramount; a 1 mg/mL solution means 0.1 mL (10 units on a U-100 insulin syringe) equals 100 mcg. Always double-check calculations with a pharmacist or medical professional.
Long-Term Storage Strategy
The best practice is to reconstitute only what you will use within the recommended shelf-life and store the rest lyophilized. Lyophilized peptides, when stored frozen, dry, and dark, can remain stable for years. Once reconstituted, the clock starts ticking. Implement an aliquoting and labeling system from day one. A simple spreadsheet tracking peptide name, lot number, reconstitution date, concentration, solvent, and discard date is invaluable for a serious user.
Conclusion: Precision, Patience, and Protocol
Reconstituting peptides is a simple process that demands discipline, not complexity. The core principles are immutable: sterility above all, precise calculation, correct solvent choice, and vigilant storage. There is no room for guesswork or cutting corners. By treating every step—from disinfecting your workspace to labeling the vial—as a critical part of the experiment or treatment, you protect your investment, ensure data integrity, and most importantly, maintain safety. Remember, the goal is to return the peptide to a solution state that mirrors its intended, pure, bioactive form. Master these steps, and you unlock the full potential of these remarkable molecular tools, whether in the pursuit of scientific discovery or personal wellness. When in doubt, consult the manufacturer's documentation and err on the side of caution. Your future self, and your results, will thank you.