Why Does Rain Make Your Car Look Dirty? Understanding Acid Rain And Its Effects
Have you ever noticed that after a rainstorm, your car often looks worse than before? Instead of being clean and shiny, it's covered in spots, streaks, and a dull film that seems impossible to remove. This phenomenon isn't just your imagination - it's a real issue that affects millions of drivers worldwide. But why does rain make your car look dirty, and what can you do about it?
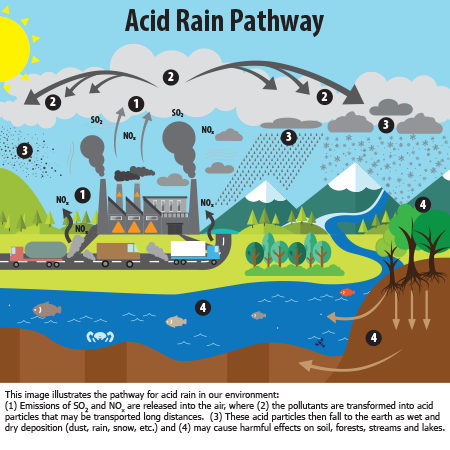
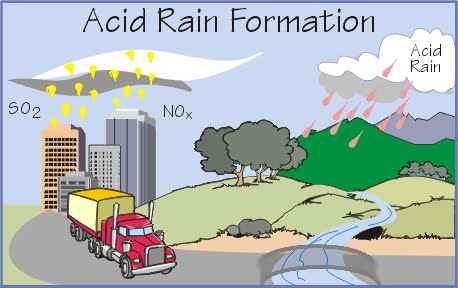
The answer lies in what we call "acid rain" - a term that might sound dramatic but accurately describes the chemical composition of modern precipitation. When rain falls through our polluted atmosphere, it picks up various contaminants including sulfur dioxide, nitrogen oxides, and other pollutants. These chemicals react with water vapor to create acidic compounds that fall to earth as rain.
When this contaminated rain hits your car's surface, it doesn't simply wash away dirt - it actually leaves behind mineral deposits, chemical residues, and acidic compounds that bond to your vehicle's paint. As the water evaporates, these substances remain, creating the cloudy, spotty appearance that frustrates so many car owners. Understanding this process is the first step toward protecting your vehicle and maintaining its appearance despite the challenges posed by modern environmental conditions.
- Leaked How To Make A Ribbon Bow So Nude Its Banned Everywhere
- Joseph James Deangelo
- Skin Club Promo Code
What Causes Acid Rain and How It Affects Your Vehicle
Acid rain forms when industrial emissions, vehicle exhaust, and other pollutants enter the atmosphere and combine with water vapor. The primary culprits are sulfur dioxide (SO2) and nitrogen oxides (NOx), which are released from power plants, factories, and vehicles. These compounds rise into the atmosphere where they react with water, oxygen, and other chemicals to form sulfuric and nitric acids.
When acid rain falls on your car, several damaging processes occur simultaneously. First, the acidic nature of the water can begin to etch into your car's clear coat and paint, especially if left to dry on the surface. Second, the rain carries with it particulate matter from the atmosphere - dust, pollen, soot, and other pollutants that settle on your vehicle's surface. Third, as the water evaporates, it leaves behind concentrated mineral deposits and chemical residues.
The pH level of acid rain typically ranges from 4.0 to 5.5, compared to pure water which has a neutral pH of 7.0. While this might not seem like a dramatic difference, automotive paint and clear coats are sensitive to pH levels, and repeated exposure to acidic compounds can cause micro-abrasions, discoloration, and a loss of shine over time. Areas with heavy industrial activity or high traffic congestion often experience more severe acid rain effects on vehicles.
- Elijah Schaffers Sex Scandal Leaked Messages That Will Make You Sick
- Twitter Porn Black
- Nude Photos Of Jessica Mann Leaked The Truth Will Blow Your Mind
The Science Behind Water Spots and Mineral Deposits
The spots you see on your car after rain aren't just dirt - they're mineral deposits left behind when water evaporates. These deposits, primarily composed of calcium and magnesium, come from both the rainwater itself and from dust and pollution particles that the rain carries. When water containing these minerals evaporates from your car's surface, it leaves behind concentrated deposits that bond to the paint.
The process is similar to what happens when you leave a glass of tap water on a wooden table - when the water evaporates, it leaves a ring. On your car's surface, these mineral deposits create a rough texture that scatters light rather than reflecting it smoothly, resulting in that dull, dirty appearance. In areas with hard water or high pollution levels, these deposits can be particularly stubborn and may require special cleaning products to remove effectively.
Temperature plays a crucial role in how these deposits form. When rain falls on a warm car surface, the water evaporates more quickly, giving minerals less time to run off before they're deposited. This is why you might notice more severe spotting on sunny days following rain, or why parking in direct sunlight after a storm can lead to worse water spots than parking in the shade.
Common Misconceptions About Rain and Car Cleaning
Many car owners believe that rain naturally cleans their vehicles, but this is actually a misconception. While rain can wash away loose surface dirt, it often leaves your car looking worse than before. This happens because rain redistributes dirt and contaminants across your car's surface rather than removing them completely. The water acts as a carrier, moving particles around and then depositing them in new locations as it evaporates.
Another common myth is that you don't need to wash your car after it rains because the rain has already "cleaned" it. In reality, the longer acidic compounds and mineral deposits remain on your car's surface, the more damage they can cause. These substances can begin to etch into the clear coat within hours, especially in warm conditions. Waiting days or weeks between washes allows these contaminants to accumulate and bond more strongly to the paint surface.
Some people also believe that all rain is the same, but this isn't true. Rain in urban areas typically contains more pollutants than rain in rural areas. Coastal regions might experience different types of mineral deposits due to sea spray mixing with precipitation. Understanding these variations can help you better protect your vehicle based on your specific location and environmental conditions.
Best Practices for Protecting Your Car from Acid Rain Damage
The most effective way to protect your car from acid rain damage is through regular washing and proper maintenance. Washing your car within 24-48 hours after rain exposure helps remove acidic compounds and mineral deposits before they can cause lasting damage. Use a pH-neutral car shampoo and clean, soft microfiber towels to avoid introducing new scratches while removing contaminants.
Applying a quality wax or paint sealant creates a protective barrier between your car's paint and environmental contaminants. These products work by creating a hydrophobic surface that causes water to bead up and roll off rather than allowing it to spread out and evaporate on the paint. Many modern ceramic coatings offer even better protection, with some lasting several years when properly maintained.
Parking strategies can also significantly reduce acid rain exposure. Whenever possible, park in covered areas or use a car cover. If covered parking isn't available, try to avoid parking under trees where sap and bird droppings can combine with rain to create even more damaging compounds. During particularly acidic rain events, which often follow periods of dry weather when pollutants have accumulated in the atmosphere, consider keeping your car garaged if possible.
Professional Solutions for Severe Acid Rain Damage
When acid rain damage becomes severe or widespread, professional detailing services may be necessary. Professional detailers have access to specialized products and equipment that can safely remove bonded contaminants without damaging your car's paint. Clay bar treatments, for example, can remove surface contaminants that regular washing can't address. For more severe etching or discoloration, paint correction services using machine polishers can restore the finish.
Some professional detailers offer decontamination services specifically designed to address acid rain damage. These services typically involve a multi-step process that includes chemical decontamination to break down and remove bonded contaminants, followed by mechanical decontamination to physically remove remaining particles. The process concludes with paint correction and protection to restore and maintain the finish.
For cars with extensive clear coat damage from years of acid rain exposure, more intensive restoration might be necessary. This could include wet sanding to remove severely damaged clear coat layers, followed by polishing and reapplication of protective coatings. While these services represent a significant investment, they can restore your car's appearance and protect it from further damage for years to come.
Long-Term Effects of Acid Rain on Different Car Surfaces
Acid rain affects various car surfaces differently, and understanding these variations can help you provide targeted protection. Clear coat and paint are most vulnerable to etching and discoloration, as the acidic compounds can break down the chemical structure of these materials over time. This damage often appears as a cloudy or hazy appearance that can't be removed with normal washing.
Metal surfaces, particularly chrome and aluminum trim, can experience oxidation and corrosion when exposed to acidic compounds over extended periods. The acid can break down protective clear coats and chrome plating, leading to pitting and discoloration. Wheels are especially susceptible since they're constantly exposed to road grime, brake dust, and acidic precipitation.
Glass surfaces can also be affected, though typically less severely than painted surfaces. Acid rain can cause water spots and light etching on windows and mirrors, which can impair visibility and create a constant film that's difficult to clean. Windshield wiper blades can deteriorate more quickly when exposed to acidic compounds, leading to streaking and reduced effectiveness during subsequent rain events.
Environmental Factors That Influence Acid Rain Severity
The severity of acid rain and its effects on your vehicle can vary significantly based on environmental factors and geographic location. Industrial areas with high concentrations of factories and power plants typically experience more acidic precipitation due to higher levels of sulfur dioxide and nitrogen oxide emissions. Urban areas with heavy traffic congestion also see increased acid rain severity due to vehicle emissions.
Geographic features can influence how acid rain affects your area. Regions downwind from industrial centers often receive more acidic precipitation as pollutants are carried by prevailing winds. Mountainous areas might experience different effects as rain passes over various terrain types, picking up different minerals and pollutants along the way. Coastal regions might deal with the added challenge of salt spray mixing with acidic compounds, creating particularly corrosive conditions.
Seasonal variations also play a role in acid rain severity. Summer months often see more severe effects due to higher temperatures accelerating chemical reactions and increasing the rate of water evaporation, which concentrates acidic compounds on vehicle surfaces. Spring and fall might bring different challenges as changing temperatures and increased biological activity in the atmosphere affect the chemical composition of precipitation.
DIY Solutions for Removing Acid Rain Spots and Deposits
For car owners who prefer to address acid rain damage themselves, several DIY solutions can be effective for mild to moderate contamination. A simple vinegar solution (one part white vinegar to one part distilled water) can help dissolve mineral deposits and neutralize acidic compounds. Apply the solution with a soft microfiber cloth, allow it to sit for a few minutes, then rinse thoroughly and dry with a clean microfiber towel.
Commercial acid rain removers are also available and can be more effective than household solutions for stubborn deposits. These products typically contain mild acids that safely dissolve mineral deposits without damaging your car's paint. Always follow the manufacturer's instructions carefully and test the product on a small, inconspicuous area first to ensure compatibility with your vehicle's finish.
For extremely stubborn spots, a clay bar treatment can be highly effective. Detailing clay picks up bonded contaminants that washing alone can't remove. Use the clay with a proper lubricant, working in small sections until the surface feels smooth. After clay barring, always apply a fresh coat of wax or sealant to protect the now-exposed paint surface.
The Role of Car Wax and Sealants in Acid Rain Protection
Quality car wax and paint sealants play a crucial role in protecting your vehicle from acid rain damage. These products create a sacrificial barrier between your car's paint and environmental contaminants, meaning that the wax or sealant takes the damage rather than your clear coat. Most quality waxes provide protection for 2-3 months, while paint sealants can last 6-12 months or longer.
The hydrophobic properties of these products are particularly valuable in acid rain protection. By causing water to bead up and roll off the surface, they reduce the contact time between acidic compounds and your car's paint. This not only helps prevent etching but also reduces the formation of water spots as there's less water left to evaporate on the surface.
Modern ceramic coatings offer even more advanced protection, with some products claiming to last for several years. These coatings chemically bond to your car's paint, creating a much harder and more durable barrier than traditional waxes or sealants. While more expensive initially, ceramic coatings can be cost-effective over time due to their longevity and superior protection against acid rain and other environmental contaminants.
When to Seek Professional Help for Acid Rain Damage
While many acid rain issues can be addressed through regular maintenance and DIY solutions, certain situations warrant professional intervention. If you notice widespread etching, discoloration, or a consistently hazy appearance that doesn't improve with thorough cleaning, it's time to consult a professional detailer. These symptoms often indicate that acidic compounds have begun to damage the clear coat itself, requiring specialized equipment and techniques to repair.
Professional help is also advisable if you're dealing with extremely hard water deposits or if DIY cleaning attempts have left your car's finish looking worse. Some mineral deposits can be so stubborn that they require professional-grade chemicals or machine polishing to remove safely. Additionally, if you're unsure about the severity of the damage or the appropriate cleaning methods for your specific vehicle, a professional assessment can prevent accidental damage from improper cleaning techniques.
Consider professional services if you're preparing to sell your car or if you want to restore the finish to like-new condition. Professional detailers can often achieve results that are difficult or impossible to replicate at home, potentially increasing your vehicle's value and making future maintenance easier by starting from a properly corrected surface.
Conclusion: Taking Control of Acid Rain's Impact on Your Vehicle
Understanding the relationship between rain and car dirtiness empowers you to take effective action against acid rain damage. By recognizing that rain itself isn't cleaning your car but rather depositing harmful compounds on its surface, you can implement a proactive maintenance strategy that preserves your vehicle's appearance and value.
The key to success lies in consistent, preventive care rather than reactive cleaning. Regular washing, proper protection with quality waxes or sealants, and strategic parking can dramatically reduce acid rain's impact on your vehicle. When damage does occur, addressing it promptly prevents minor issues from becoming major problems that require expensive professional intervention.
Remember that your car represents a significant investment, and protecting it from environmental damage is an important part of vehicle ownership. By understanding the science behind acid rain and implementing the protection strategies outlined in this article, you can keep your car looking its best despite the challenges posed by modern environmental conditions. The effort you invest in protecting your vehicle today will pay dividends in maintained appearance, preserved value, and reduced long-term maintenance costs.